
Scientific abstract
M. Schlaminger J. Hindinger
Download
BACKGROUND/AIM
Urinary electrolytes are used to aid the diagnosis hypovolemia, kidney damage, acid-base, and electrolyte disorders. Quantification of urinary sodium (Na), chloride (Cl), and potassium (K) by ion-sensitive electrodes (ISE) is a widely used for the management of critically ill patients. [1], [2] Established methods require pre-analytical dilution steps, which entail metrological disadvantages. The aim of the study is to quantify the pre-analytical error caused by the dilution and to compare a novel undiluted urine method EXIAS e|1 Analyzer (e|1) with diluted Roche 9180 Electrolyte Analyzer (9180).
METHODS
A method comparison study (CLSI EP9A3, [3]) using random and first morning mid-stream urine samples from 145 male and female donors between the ages of 20 and 60 and different dietary habits (vegetarian, vegan, omnivore) and a precision study (CLSI EP5A3, [4]) with 3 dedicated donor pools were performed. The statistical analysis for the comparison of both methods was performed using Deming regression. Repeatability (SD1) and Reproducibility (SD2) were estimated based on ANOVA. The samples on 9180 were diluted according to the instructions for use, with 2 dilutions being prepared and compared per aliquot (as factor in SD2), additional dilution with a.dest. for K≥60mmol/L.
RESULTS
The regression analysis of the comparison study demonstrated a slope from 0.99 (Na) to 1.02 (Cl). The results correlated well (R²>0.98) and showed that e|1 and 9180 have no significant bias. It can thus be shown that the elimination of the dilution of the urine sample has no negative impact on the quantification of the analytes on the e|1. The deviations in the lower range of Na and Cl can be attributed to the uncertainty of the 9180 (x) resulting from the dilution. The precision study shows clear differences in the SD2 between e|1 and 9180, with the difference between the concentration of the patient sample and the diluent modulating the uncertainty on the 9180; e.g., Na at 44mmol/L; e|1: SD1=0.49, SD2=1.59; 9180: SD1=1.29, SD2=4.81. The significant difference between SD1 and SD2 shows that dilution is a non-negligible source of error. 25% of the samples required an additional dilution step with a.dest. due to altered K.
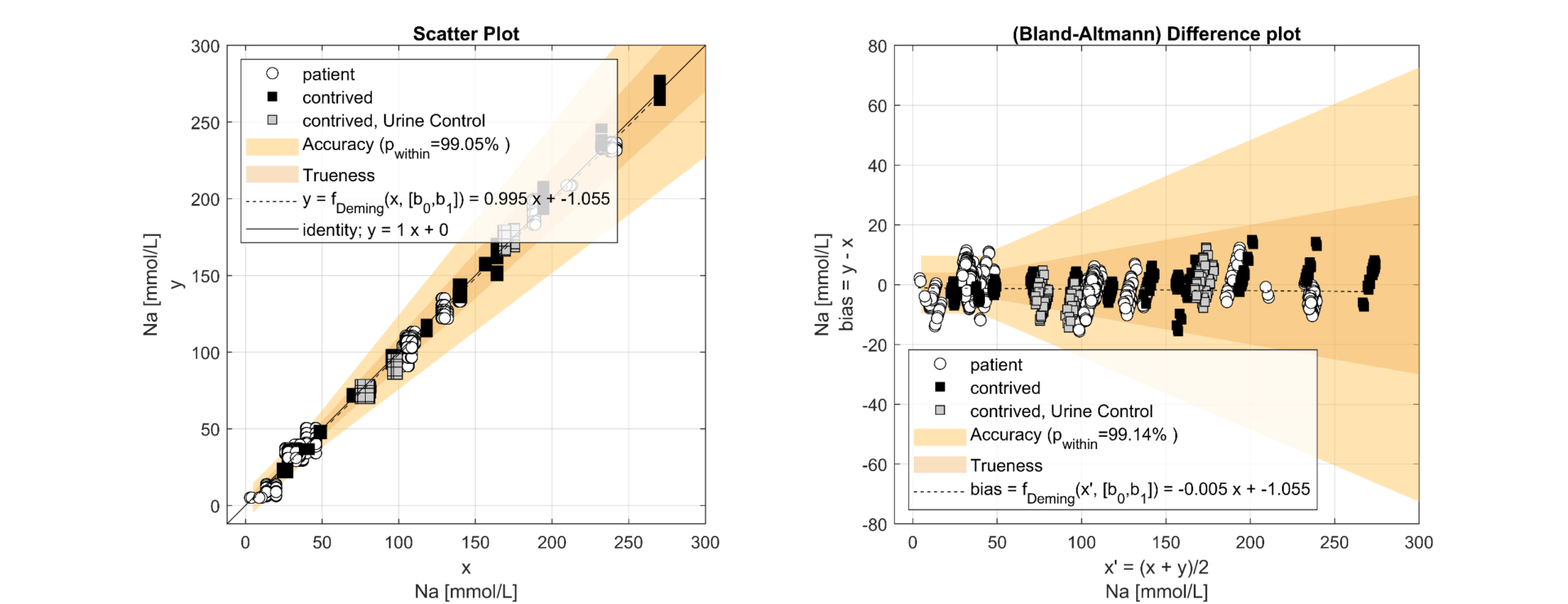
Figure 1.1 Method Comparison for Sodium according CLSI EP09A3 comparing 8 x e|1 (y, undiluted) vs. 2 x 9180 (x, diluted)
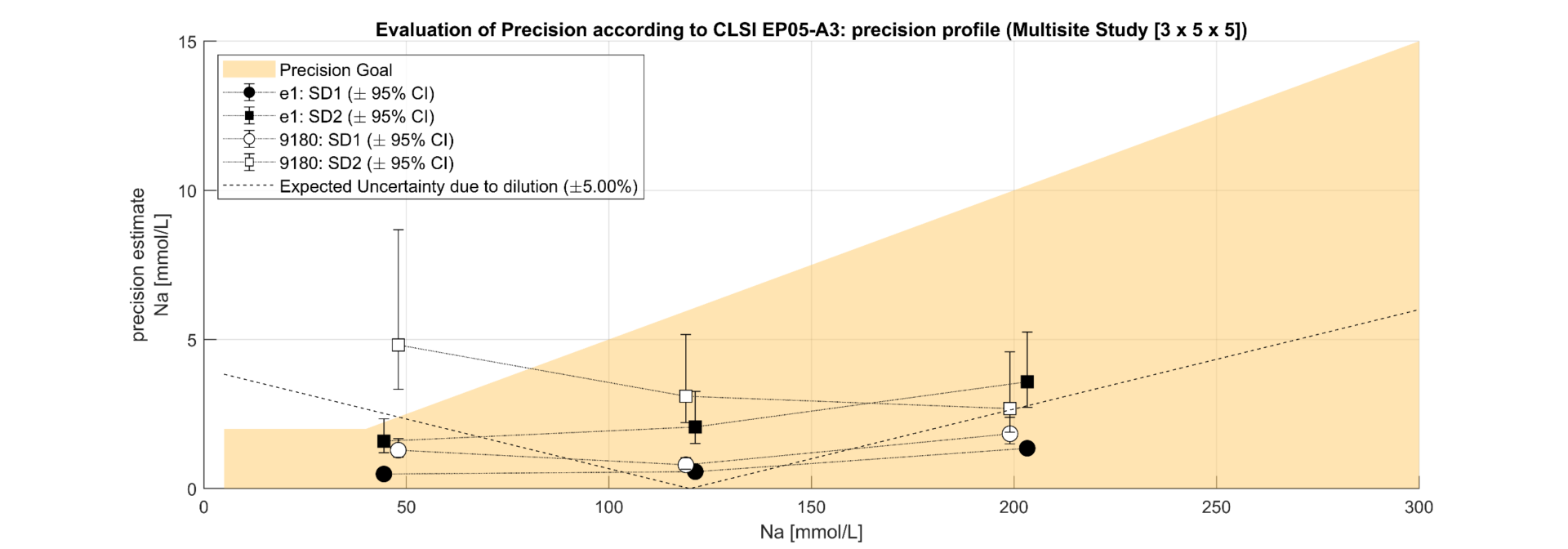
Figure 2.1. Precision profile for Sodium according CLSI EP05A3 for Repeatability SD1 and Reproducibility SD2 considering 8 x e|1 (undiluted) , respectively 2 x 9180 (diluted) as factors for SD2; expected effect of dilution error shown dashed as additional factor in SD2 to 9180
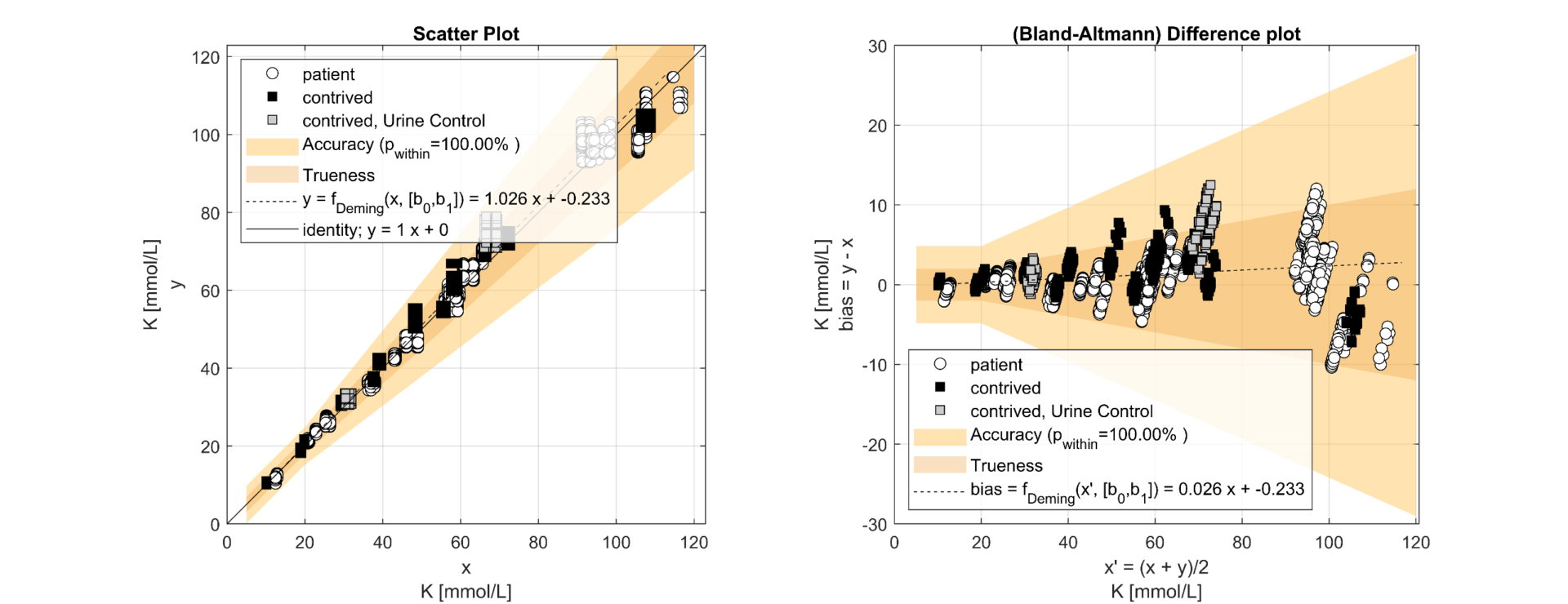
Figure 1.2 Method Comparison for Potassium according CLSI EP09A3 comparing 8 x e|1 (y, undiluted) vs. 2 x 9180 (x, diluted)

Figure 2.2. Precision profile for Potassium according CLSI EP05A3 for Repeatability SD1 and Reproducibility SD2 considering 8 x e|1 (undiluted) , respectively 2 x 9180 (diluted) as factors for SD2; expected effect of dilution error shown dashed as additional factor in SD2 to 9180
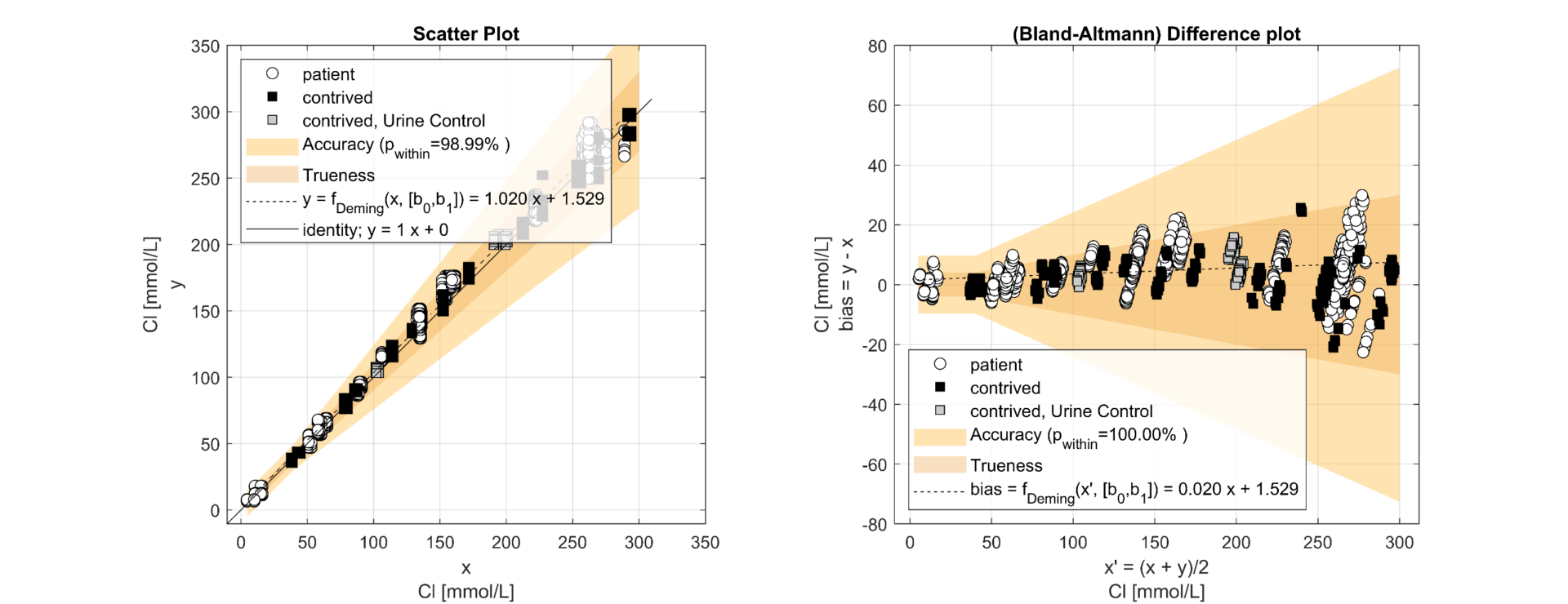
Figure 1.3 Method Comparison for Chloride according CLSI EP09A3 comparing 8 x e|1 (y, undiluted) vs. 2 x 9180 (x, diluted)
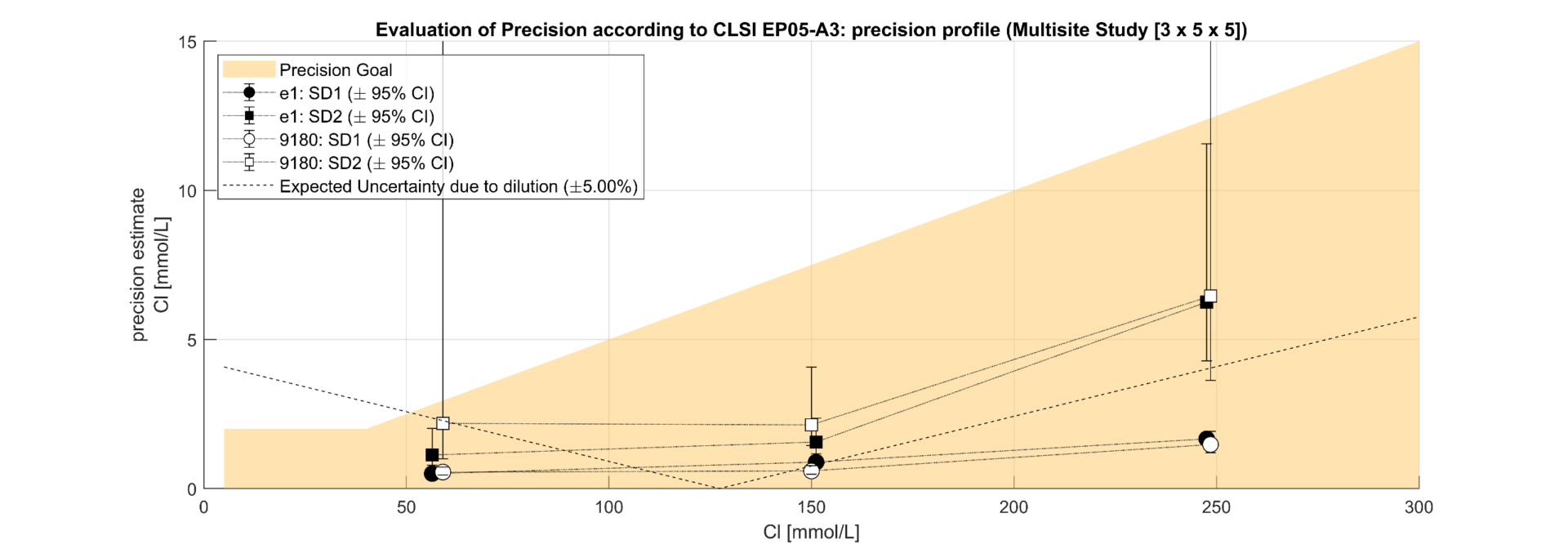
Figure 2.3. Precision profile for Chloride according CLSI EP05A3 for Repeatability SD1 and Reproducibility SD2 considering 8 x e|1 (undiluted) , respectively 2 x 9180 (diluted) as factors for SD2; expected effect of dilution error shown dashed as additional factor in SD2 to 9180
WORKFLOW COMPARISON: EXIAS e|1 ANALYZER vs. ROCHE 9180
The workflow involved in analyzing urinary electrolytes plays a crucial role in determining the efficiency and practicality of a diagnostic method.
The e|1 Analyzer workflow begins with the collection of random or first morning mid-stream urine samples. These samples are directly loaded onto the analyzer without any pre-analytical dilution steps. The analyzer then utilizes ion-sensitive electrodes (ISE) to quantify the concentrations of sodium (Na), chloride (Cl), and potassium (K) in the undiluted urine samples. The quantification process is automated, minimizing the need for manual intervention. The results obtained from the e|1 Analyzer can be directly interpreted and used for clinical decision-making.
On the other hand, the workflow of the Roche 9180 Electrolyte Analyzer [5] involves a series of pre-analytical dilution steps. After urine sample collection, the samples are diluted according to the instructions for use. Typically, two dilutions per aliquot are prepared and compared, with an additional dilution using a diluent (a.dest.) for samples with potassium concentrations equal to or greater than 60 mmol/L. Once the samples are appropriately diluted, they are loaded onto the Roche 9180 Analyzer. Similar to the e|1 Analyzer, the Roche 9180 employs ion-sensitive electrodes to quantify the concentrations of Na, Cl, and K in the urine samples. The results obtained are then analyzed and interpreted for clinical purposes.
A notable distinction between the two workflows lies in the dilution step. The e|1 Analyzer eliminates the need for dilution, allowing for direct analysis of undiluted urine samples. This streamlined approach simplifies the workflow and reduces the potential sources of error associated with the dilution process. In contrast, the Roche 9180 Analyzer requires the dilution of urine samples, introducing an additional pre-analytical step that may contribute to variability and uncertainty in the results. The study findings indicated that dilution on the 9180 Analyzer indeed led to deviations and increased uncertainty, particularly in the lower range of Na and Cl concentrations, as well as in the upper K concentrations.
Overall, the e|1 Analyzer workflow offers a more straightforward and efficient process by eliminating the pre-analytical dilution step. This feature reduces the potential for error and provides more reliable quantification of urinary electrolytes. In comparison, the Roche 9180 Analyzer workflow necessitates the extra step of dilution, which introduces an additional source of error and uncertainty.
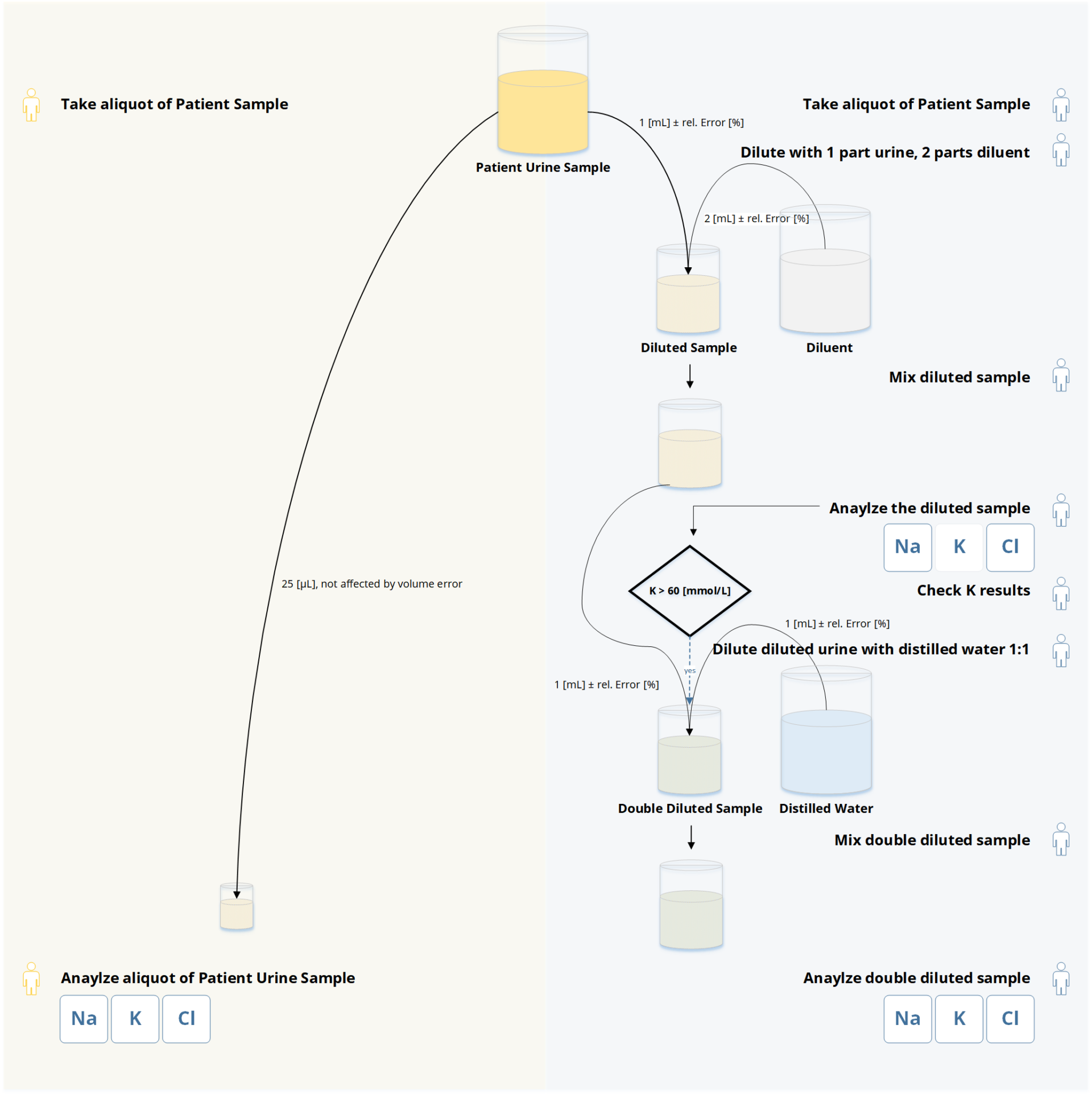
Figure 3 Workflow Comparison: EXIAS e|1 Analyzer (left) vs. Roche 9180 (right)
Conclusion
This study compared the quantification of urinary electrolytes using the novel undiluted urine method on the e|1 Analyzer with the traditional diluted method on the Roche 9180 Analyzer. The comparison study demonstrated that both analyzers showed good correlation and no significant bias, indicating that the elimination of the dilution step did not negatively impact the quantification of analytes on the e|1 Analyzer.
Moreover, the precision study revealed significant differences in the standard deviation (SD2) between the e|1 and 9180 Analyzers, suggesting that pre-analytical dilution is a notable source of error. This finding highlights the importance of considering the potential impact of dilution on the accuracy and precision of urinary electrolyte measurements.
The e|1 Analyzer offers several advantages over the established analyzers, including the elimination of pre-analytical dilution steps. This not only simplifies the workflow but also reduces the potential for error associated with dilution. Additionally, the e|1 Analyzer provides the capability to analyze undiluted urine samples directly, potentially saving time and resources in a clinical setting.
Overall, the results of this study suggest that the undiluted urine method on the e|1 Analyzer holds promise as an alternative approach for the quantification of urinary electrolytes. It has the potential to improve the accuracy and efficiency of diagnostic testing in the management of critically ill patients and the diagnosis of electrolyte disorders.
References:
[1] Umbrello et al. "Urine electrolytes in the intensive care unit: From pathophysiology to clinical practice." Anesthesia & Analgesia 131.5 (2020): 1456-1470.
[2] Seifter et.al. "Disorders of acid-base balance: new perspectives." Kidney Diseases 2.4 (2016): 170-186.
[3] Evaluation of Precision of Quantitative Measurement Procedure; Approved Guideline – Third Edition. CLSI document EP05-A3; 2014
[4] Measurement Procedure Comparison and Bias Estimation Using Patient Samples; Approved Guideline – Third Edition- CLSI document EP09-A3; 2013
[5] 9180 Electrolyte Analyzer Instructions for Use EN; Version 7.1; 2013
