IMPACT OF PLASMA PROTEIN LEVELS ON ELECTROLYTE MEASUREMENTS: A COMPARATIVE STUDY OF DIRECT AND INDIRECT ION-SELECTIVE ELECTRODE METHODS
Schlaminger¹, B. Webb², A. Bartel², T. Niedrist³, M. Herrmann³, H. Scharnagl³
1 EXIAS Medical GmbH, Graz, Austria
2 MEON Medical Solutions GmbH & Co KG, Graz, Austria
3 Clinical Institute of Medical and Chemical Laboratory Diagnostics, Medical University of Graz, Austria
BACKGROUND/AIM
Accurate electrolyte measurement is critical for diagnosing and managing disorders of fluid and electrolyte balance. This study evaluates the effect of plasma protein levels on sodium (Na), potassium (K), and chloride (Cl) measurements using direct and indirect ion-selective electrode (ISE) methods, focusing on protein-induced matrix effects such as pseudohypernatremia and pseudohyponatremia. These phenomena occur because indirect ISE methods measure ion activity in a diluted sample, requiring mathematical correction based on assumptions about plasma water content that may not hold in abnormal protein states [1]. Previous research demonstrated that up to 25% of samples from critically ill patients could be misclassified when using indirect ISE methods in cases of abnormal protein concentrations [1] [2], of which 97% occur in hypoproteinemic samples [2].
METHODS
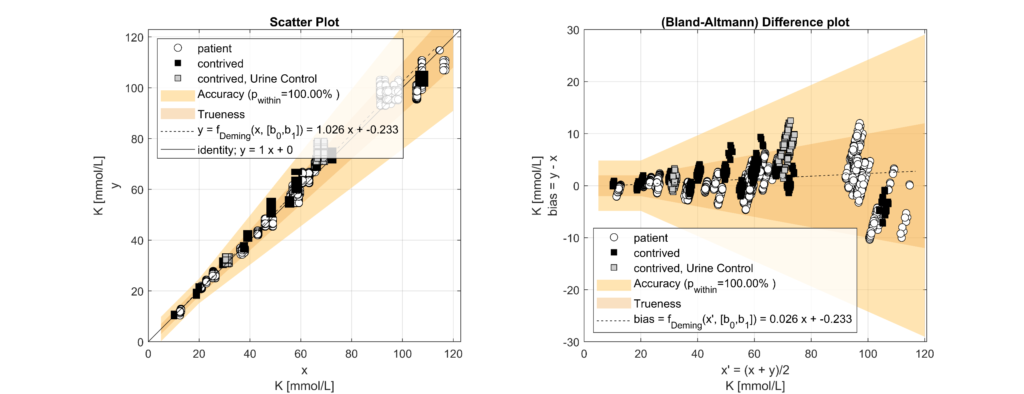
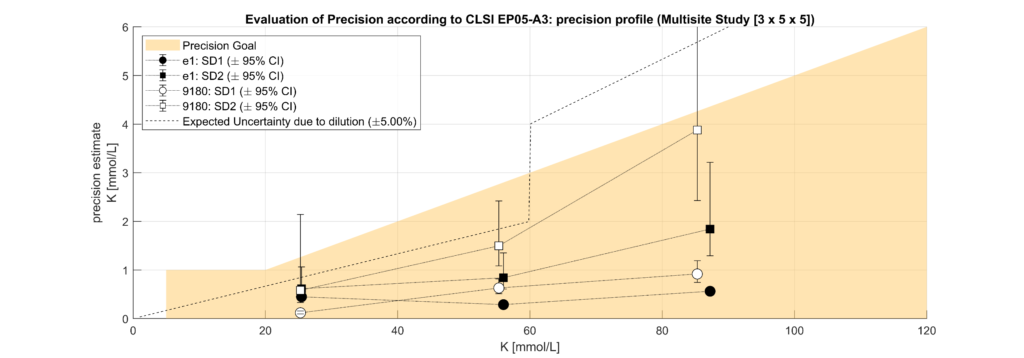
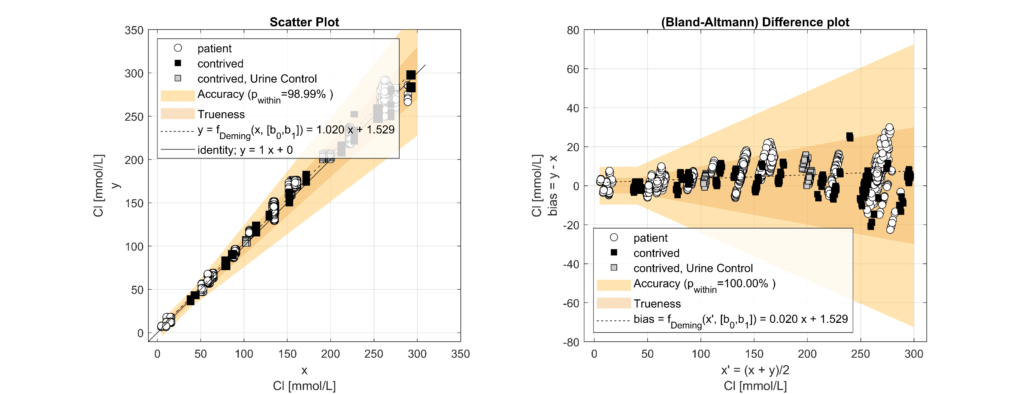
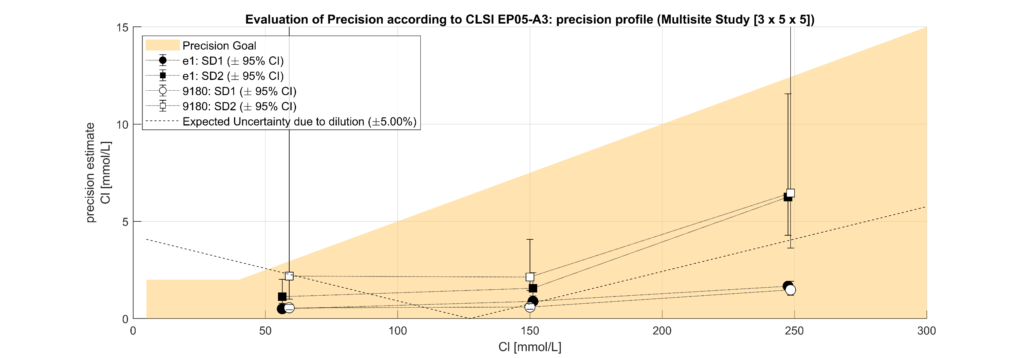
In leftover plasma samples (n=120, Li Hep), electrolyte (Na, K, Cl) concentrations were measured on the solite analyzer (MEON Medical Solutions) with integrated m|1 electrolyte module (EXIAS Medical) as well as on the Cobas 8000 analyzer (Roche Diagnostics). The m|1 electrolyte module uses direct ISE, while the Cobas 8000 uses indirect ISE. Additionally, total protein (TP) concentrations were determined for the same samples on the Cobas 8000.
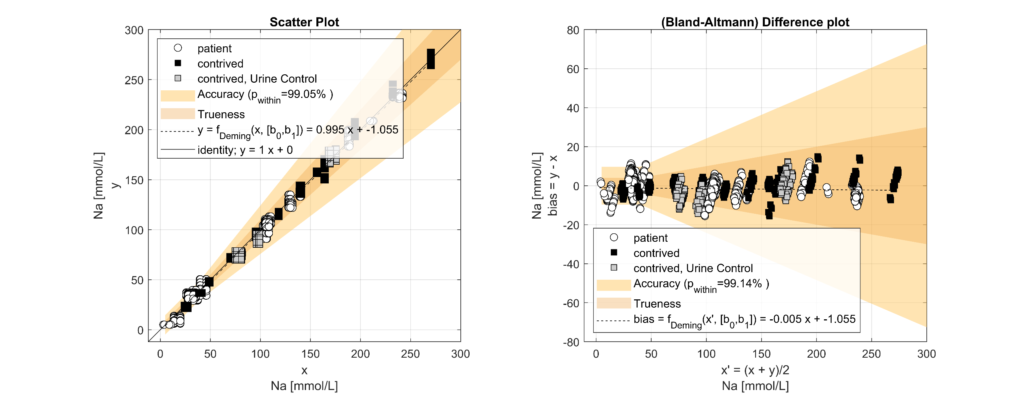
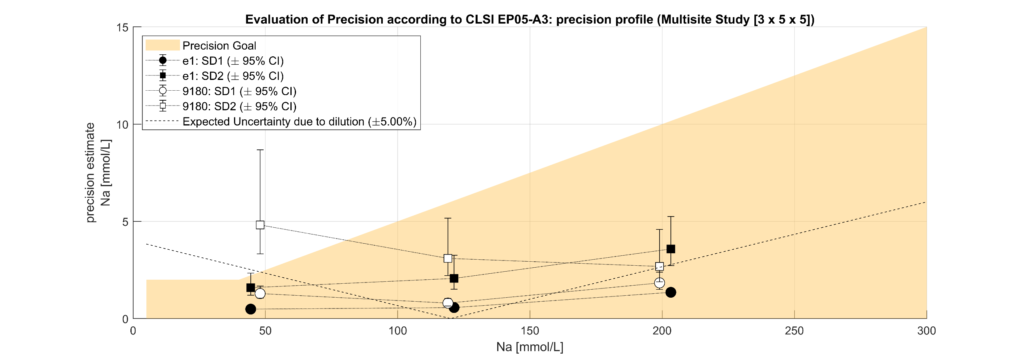
Samples were classified into hypoproteinemic (<6.6 g/dL; 64.4%) and normoproteinemic (6.6–8.7 g/dL; 35.6%) groups based on TP levels. Systematic biases between methods were assessed using Bland-Altman analysis and linear regression modelling. A multifactorial linear model incorporating electrolyte concentration (Na, K, Cl), TP levels, and an intercept was used to quantify the impact of protein levels on electrolyte measurements.
RESULTS
Significant biases (p < 0.05) were observed across all electrolytes. For every 1 g/dL increase in TP, indirect ISE underestimated Na by 0.77 mmol/L (p < 0.001), K by 0.05 mmol/L (p < 0.001) and Cl by 1.02 mmol/L (p < 0.001) compared to direct ISE. Hence, indirect ISE overestimates Na levels (e.g., pseudohypernatremia) in hypoproteinemic specimens and underestimates Na levels (e.g., pseudohyponatremia) in hyperproteinemic specimens [3].
These observed biases closely matched theoretical expectations based on plasma water fraction changes with varying protein concentrations [4]. Correcting for systematic differences confirmed that biases were exclusively attributable to TP levels.
Notably, our dataset did not include hyperproteinemic samples (>8.7 g/dL), limiting our direct observations to hypoproteinemic and normoproteinemic ranges.



Conclusion
Direct ISE methods provide more accurate measurements of Na, K, and Cl across varying protein levels (3.7 – 8.1 g/dL) compared to indirect ISE methods, which are prone to protein-induced matrix effects due to their dilution step and the compensation applied to account for it. The magnitude of this effect is clinically significant, particularly for sodium measurements in critically ill patients who are often present with abnormal protein levels.
Our results confirm and extend earlier observations by Dimeski et al. [1],[2], highlighting that the risk of misclassification is substantial and directly proportional to the deviation from normal protein levels. The observed coefficient of -0.77 mmol/L per g/dL for sodium aligns well with previous estimates (-0.78 to -0.98 mmol/L per g/dL) [2], supporting the consistency of this phenomenon across different analytical platforms.
These findings align with recent consensus recommendations suggesting direct ISE methods should be preferred [5]. Selecting appropriate measurement techniques is critical in clinical settings to minimize errors in electrolyte assessment caused by plasma protein variations. [5], [6]
Referenzen:
[1] Dimeski G, Barnett RJ. Effects of total plasma protein concentration on plasma sodium, potassium and chloride measurements by an indirect ion selective electrode measuring system. Crit Care Resusc. 2005;7(1):12-15.
[2] Dimeski G, Morgan TJ, Presneill JJ, Venkatesh B. Disagreement between ion selective electrode direct and indirect sodium measurements: estimation of the problem in a tertiary referral hospital. J Crit Care. 2012;27(3):326.e9-16.
[3] Liamis G, Liberopoulos E, Barkas F, Elisaf M. Spurious electrolyte disorders: a diagnostic challenge for clinicians. Am J Nephrol. 2013;38(1):50-57.
[4] Nguyen MK, Ornekian V, Butch AW, Kurtz I. A new method for determining plasma water content: application in pseudohyponatremia. Am J Physiol Renal Physiol. 2007;292(5)
[5] Langelaan ML, Kamp L, Zandijk E, Raijmakers MT. Prevalence of pseudonatremia in a clinical laboratory - role of the water content. Clin Chem Lab Med. 2017 Mar 1;55(4):546-553
[6] King RI, Mackay RJ, Florkowski CM, Lynn AM. Electrolytes in sick neonates - which sodium is the right answer? Arch Dis Child Fetal Neonatal Ed. 2013;98(1)